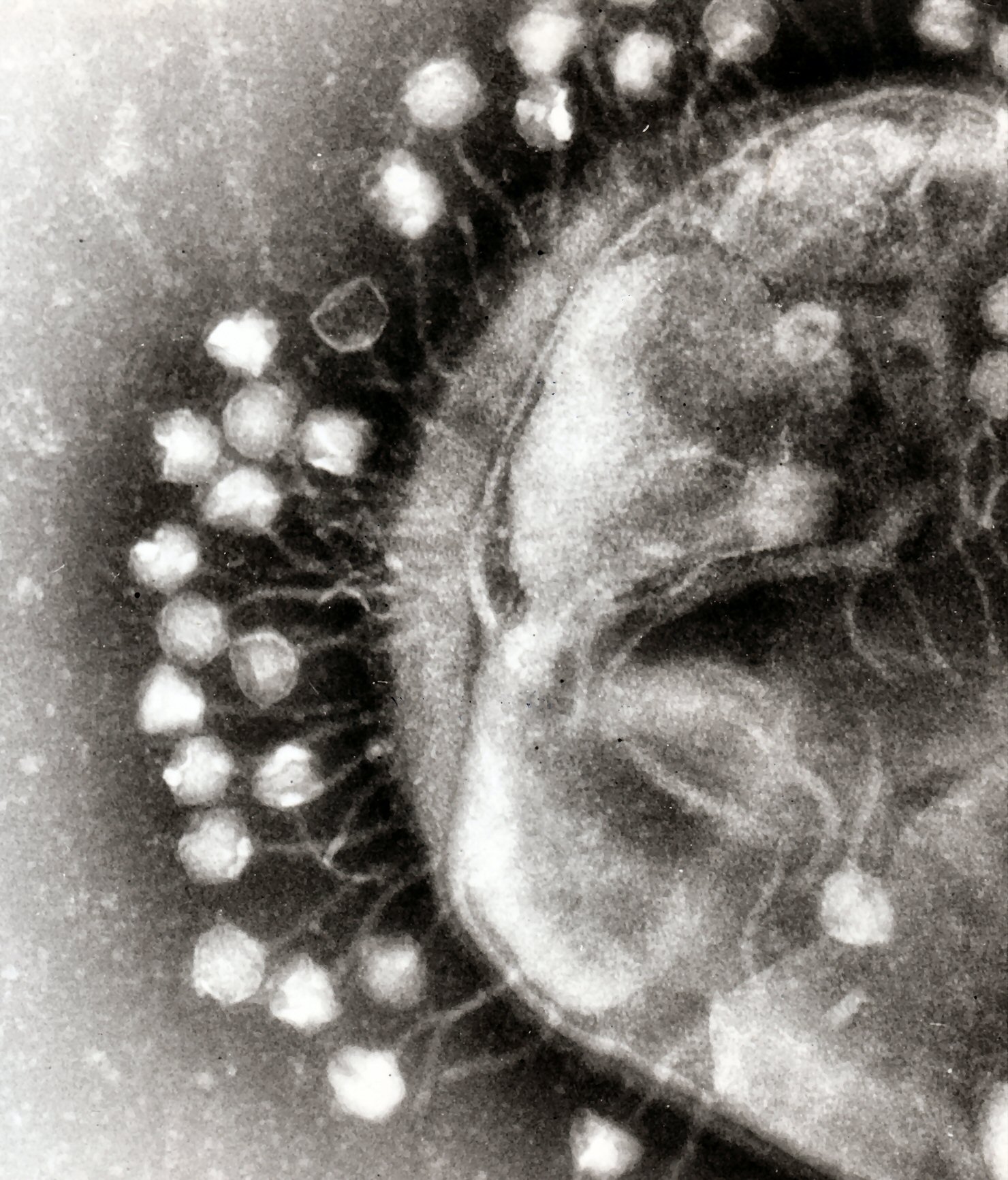
Microbial communities are elegantly complex, with all members competing, cooperating, or simply co-existing with all other members. These interactions are especially evident between bacteria and bacteriophages. In many cases, bacteriophages interact with bacterial populations by destroying bacteria through predation [1], by mediating gene transfer through transduction [2], or by controlling gene expression of their bacterial hosts [3]. Some phages integrate into bacterial chromosomes (prophages), which can confer benefits by preventing further phage infection or modulating gene expression, but this can also make the bacteria host susceptible to death by phage induction (process by which DNA damage stimulates excision of the phage genome from the bacterial chromosome, which is followed by phage assembly and release while destroying the bacteria). Some bacteria have developed methods to obtain competitive advantages over prophage containing competitors (lysogenic bacteria) by killing them through stimulation of phage induction, such as those reported by Selva et al [4].
In their report, Selva et al studied the competitive interactions between Streptococcus pneumonia (a bacterial pathogen targeted by pneumococcal vaccine) and Staphylococcus aureus (an opportunistic pathogen). In their study, they report how relatively low levels of hydrogen peroxide are produced by S. pneumonia with the intent of stimulating DNA damage in their S. aureus competitors, thereby promoting phage induction and consequent destruction of the lysogenic bacteria. This is thought to be a particularly effective strategy because S. aureus bacteria commonly maintain prophages.
In their study, the researchers first showed that lysogenic strains of S. aureus are killed when exposed to levels of hydrogen peroxide often found in S. pneumonia cultures. The non-lysogenic bacteria were not affected by the treatment of hydrogen peroxide, thereby supporting the selective lethality of hydrogen peroxide in lysogenic bacteria. Not only did the group show that lysogenic S. aureus is more susceptible to hydrogen peroxide lethality, but they also showed that phages are produced is greater quantities when the cultures are treated with hydrogen peroxide. S. pneumonia was resistant to the induction effects whether it was lysogenic or not, and co-culture with S. pneumonia and S. aureus resulted in phage induction and destruction of the lysogenic S. aureus strains. The authors made the point that S. aureus produces an effective catalase that protects it from hydrogen peroxide toxicity, suggesting it should be resistant to the effects of hydrogen peroxide. Interestingly, while catalase protects non-lysogenic S. aureus from destruction by hydrogen peroxide, it was significantly less able to protect lysogenic S. aureus from death. The authors speculate that this occurs because significantly more hydrogen peroxide is required to kill a bacterium, compared to that required to stimulate the SOS response. I think this also further emphasizes the potential competitive disadvantage of harboring prophages, because they can make their host susceptible to antimicrobial insults that they would otherwise be resistant to. This overall effect of prophages on antimicrobial susceptibility was also shown to occur upon antibiotic treatment, in which lysogenic bacteria were more susceptible to prophage inducing antibiotics, compared to non-lysogenic bacteria.
This study is interesting for many reasons. First, this study illustrates the importance of bacteriophages in bacterial interactions. Phages can often be overlooked when considering interactions between bacteria, but they are intricately involved in many bacterial processes and deserve close attention. Second, as illustrated by this paper, insight into how phages affect bacterial susceptibility to antimicrobial chemicals, like hydrogen peroxide, can provide a greater understanding of how they might interact with similar chemicals, like antibiotics. Third, because these effects on phage induction can affect the rates of phage mediated gene transfer (transduction), including antibiotic resistance genes and virulence factors, this study illustrates the importance of these interactions in bacterial virulence and antibiotic resistance transduction (addressed somewhat by the authors, but further studies are required). I think this also suggests the importance of understanding interactions among entire bacterial/phage communities, in addition to closed culture-based systems, because there are so many interactions that affect each other directly and indirectly. Finally, the authors mention the potential implications of this work in pneumococcal vaccination, which may alter the effects of S. aureus gene expression, abundance, and transduction, as a result of removing the S. pneumonia population.
I think this last topic of vaccination implications deserves an entire discussion on its own, so I am going to defer this until next time. Additionally, I want to point out that is is a somewhat old article, but I think it is still a very interesting story, a good read, and a study worth discussing. The article is free to read to check it out by following the reference link below.
Works Cited
1. Rodriguez-Valera F, Martin-Cuadrado AB, Rodriguez-Brito B, Pasic L, Thingstad TF, Rohwer F, Mira A. Explaining microbial population genomics through phage predation. Nature reviews Microbiology. 2009;7(11):828-36. Epub 2009/10/17. doi: 10.1038/nrmicro2235. PubMed PMID: 19834481.
2. Varga M, Kuntova L, Pantucek R, Maslanova I, Ruzickova V, Doskar J. Efficient transfer of antibiotic resistance plasmids by transduction within methicillin-resistant Staphylococcus aureus USA300 clone. FEMS microbiology letters. 2012;332(2):146-52. Epub 2012/05/05. doi: 10.1111/j.1574-6968.2012.02589.x. PubMed PMID: 22553940.
3. Wirtz C, Witte W, Wolz C, Goerke C. Transcription of the phage-encoded Panton-Valentine leukocidin of Staphylococcus aureus is dependent on the phage life-cycle and on the host background. Microbiology. 2009;155(Pt 11):3491-9. Epub 2009/08/08. doi: 10.1099/mic.0.032466-0. PubMed PMID: 19661179.
*Phage picture (public domain) taken from this site.
No comments:
Post a Comment